This colloidal solution was directly added to centrifugal spin filters (Amicon Ultra-4, 10k MWCO regenerated cellulose), and the nanoparticles were collected at 4000× g and washed in purified water. The nanoparticles were allowed to stabilize for at least 60 min. 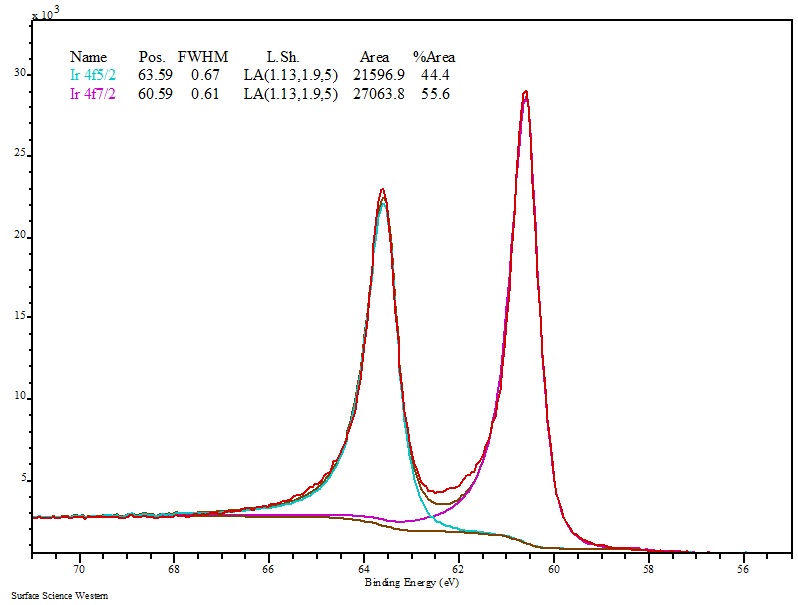
The solution gradually turned from dark brown to black over 30 min. For larger scale syntheses of 500 mL total volume, 25 mL iridium (III) chloride solution was used (diluted to 1.0 mM) and 5.0 mL borane morpholine was added (final 10 mM concentration) with rapid stirring. A solution of 1.0 M borane morpholine (Alfa Aesar) was also prepared by bath sonication. A 20 mM iridium (III) chloride (Acros Organics) stock was prepared by bath sonication and stirred for at least 20 min to generate an optically clear solution. Although iridium is a relatively expensive material like other noble metals, its current value as a commodity is approximately three quarters the price of gold and half that of rhodium, making it an interesting economic alternative.Īll synthesis reactions were performed at room temperature under aerobic conditions in purified 18 MΩ water. These novel IrNPs have not been readily explored for medical purposes despite the material’s chemical inertness and superior density. In the current study, we present the synthesis of IrNPs and their in vitro biocompatibility as well as that of iridium ions, which has not previously been evaluated in the selected cell lines. An isotope of iridium, 192Ir, is a commonly used brachytherapy gamma emitter, and part of the success of this material is due to the high density, i.e., the large number of atoms in a small volume of the material. Iridium is one of the least reactive metals, considered generally biologically compatible, and has an elemental density of 22.56 g/cm 3 (second only to osmium, which is known to be highly toxic). Here, we present a synthetic method to produce iridium nanoparticles (IrNPs), which are predicted to have strong radiation attenuation due to its high density. The majority of the chemical and engineering studies to date have focused on AuNPs to enhance radiation interactions, although bismuth and hafnium have been examined for diagnostic and therapeutic applications respectively. Therefore, functional materials that interact with radiation, such as high atomic number and high-density nanoparticles, may improve the performance of these modalities. High-energy radiation is utilized extensively in medicine including in diagnostic imaging and radiation therapy. The exceptional surface stability and known biological compatibility of these elements, as well as their potential novel physical properties on the nanoscale, make them unique alternatives to AuNPs. However, nanoparticles composed of platinoid elements, which are commonly employed for their surface catalytic properties, have yet to be thoroughly examined for biomedical applications. Gold nanoparticles (AuNPs) have been investigated extensively for sensing and therapeutic applications, while other noble metals, including silver, have found niche uses such as anti-microbials. In nanomedicine, these unique biomaterials have drawn significant attention due the ability to tailor their biological interactions through surface modifications for a wide range of applications. Noble metal nanoparticles are a mainstay of emerging nanotechnologies due to their interesting optical, electronic, and surface catalytic properties. Overall, these results support the potential for the in vivo application of this nanomaterial. Furthermore, the IrNPs were assessed in a hemolytic assay and found to have no significant impact on red blood cells when exposed to concentrations up to 100 μM. In vitro metabolic toxicity assays using hepatocyte and macrophage cells demonstrate that both IrNPs and iridium(III) chloride are well tolerated at concentrations of up to 10 μM iridium. These nanoparticles are highly crystalline as observed by X-ray diffraction and high-resolution transmission electron microscopy (TEM). Unlike other solution-based synthesis methods, uniform and monodispersed IrNPs are produced without the use of surfactants or other solubilizing ligands. We have developed an aqueous synthetic method to produce small (~ 2 nm) iridium nanoparticles (IrNPs) by reduction of iridium(III) chloride using a borohydride reducing agent. High-density inorganic nanoparticles have shown promise in medical applications that utilize radiation including X-ray imaging and as radiation dose enhancers for radiotherapy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
